Offres
POUR ÊTRE COMPÉTITIVE SUR LE MARCHÉ MONDIAL SURCHARGÉ D’AUJOURD’HUI, VOTRE ENTREPRISE DOIT OFFRIR DES PRODUITS ET DES SERVICES SÛRS ET DE QUALITÉ SUPÉRIEURE. MAIS SE CONFORMER AUX RÉGLEMENTATIONS DU SECTEUR ET RESTER COMPÉTITIF ET DURABLE SIGNIFIE RELEVER DES DÉFIS. QU’IL S’AGISSE DE LA NÉCESSITÉ DE RATIONALISER LES PROCESSUS POUR RÉDUIRE LES COÛTS OU DES PRESSIONS EXERCÉES POUR RÉDUIRE L’UTILISATION DE L’ÉNERGIE, DE L’EAU ET DES RESSOURCES, CHAQUE ENTREPRISE S’ATTEND À MAXIMISER L’EFFICACITÉ ET LA VALEUR DES INVESTISSEMENTS EN MATÉRIEL, LOGICIELS, MATIÈRES PREMIÈRES ET RESSOURCES. ET QUEL QUE SOIT VOTRE DOMAINE, VOUS AVEZ BESOIN DE L’ASSURANCE DE LA TRANSPARENCE, DE LA TRAÇABILITÉ ET DE LA CONFORMITÉ DES PROCESSUS DANS L’ENSEMBLE DE VOTRE ORGANISATION.
NOS SOLUTIONS
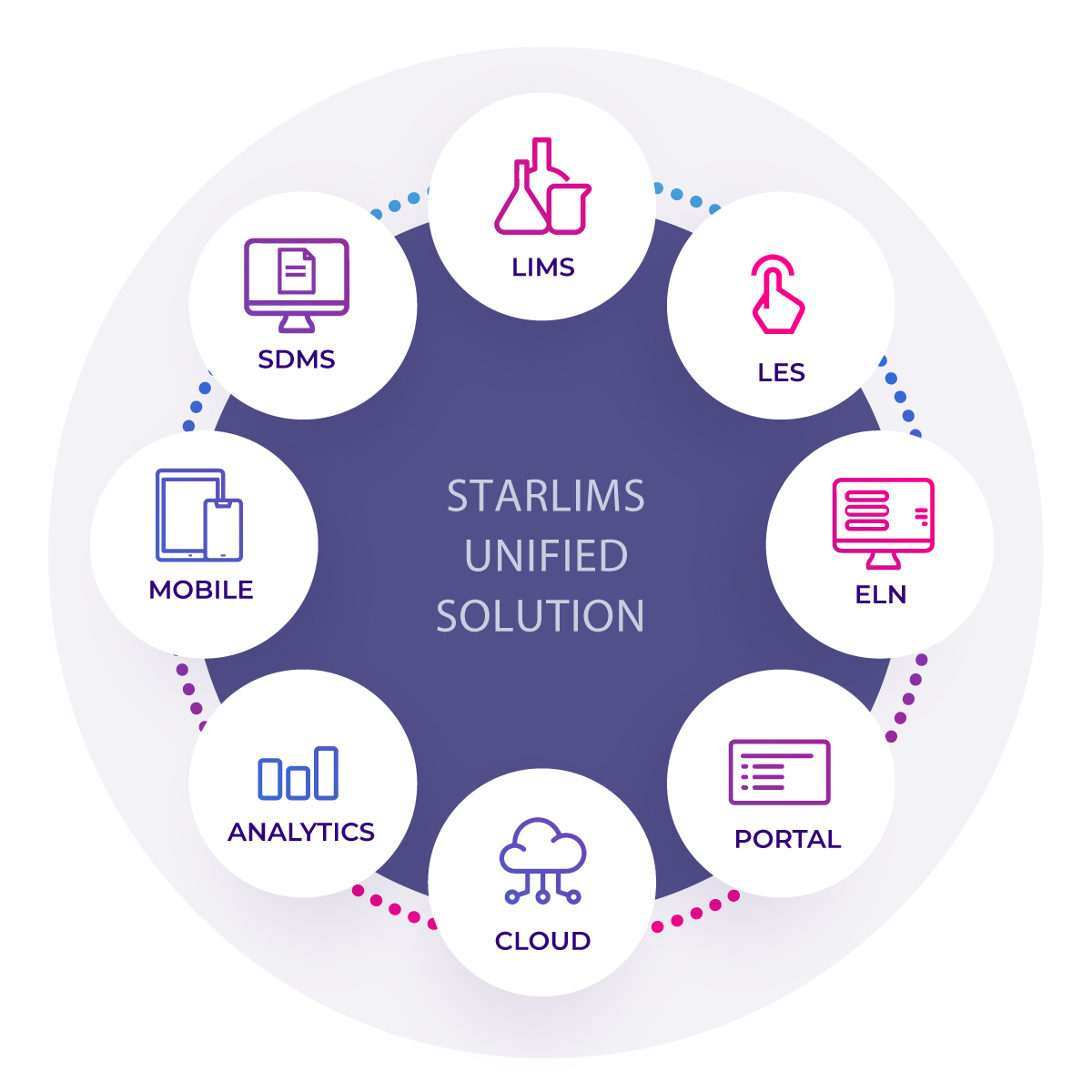
LIMS
Traitez l’ensemble du cycle de vie des produits avec le système de gestion des informations de laboratoire
(LIMS).
SDMS
Gérer vos documents et vos données avec le Scientific Data Management System (SDMS) (système de gestion de données scientifiques).
ELN
ELN est une solution électronique puissante à même d’optimiser l’efficacité de votre laboratoire et d’assurer votre conformité aux réglementations en vous permettant d’exécuter, de façon ininterrompue, vos processus d’analyses.
LES
Avec le système STARLIMS Laboratory Execution System (LES), les laboratoires peuvent facilement documenter les étapes importantes de l’exécution d’une méthode au moment où elles sont réalisées (exécution en laboratoire).
Analyses Avancées
Obtener les perspectives dont vous avez besoin pour prendre de meilleures décisions.
Mobile
Accéder à vos données de laboratoire pratiquement n’importe où, sur smartphone ou tablette.
Services Cloud
Déployer votre solution LIMS dans un environnement sécurisé, sans subir les coûts de maintenance d’une infrastructure interne.
COMPOSEZ VOTRE SOLUTION
La solution unifiée est une combinaison puissante d’offres. Elle s’appuie sur la solution LIMS, une solution de tout premier ordre. Améliorez votre système LIMS en ajoutant un produit ELN pour mettre en application l’exécution des méthodes et/ou un produit SDMS pour rationaliser la collecte de données et la gestion documentaire. Enrichissez votre pratique avec les fonctions d’Analyses Avancées et les fonctions Mobile. Le choix vous appartient.
SECTEURS
STARLIMS CONNAÎT VOS ACTIVITÉS
De la R&D à la fabrication et au-delà, STARLIMS optimise l’ensemble du cycle de vie des produits. Grâce à des fonctions et des capacités préconfigurées et spécifiques à l’industrie, les experts de STARLIMS peuvent rendre votre solution LIMS rapidement opérationnelle afin que vous puissiez vous concentrer sur la mise sur le marché plus rapide de produits sûrs et de haute qualité. Sélectionnez un secteur d’activité pour trouver les spécifications techniques et en savoir plus sur la façon dont STARLIMS aide votre organisation à exceller.
AVIS CLIENTS
“Le logiciel que nous utilisions auparavant était basé sur Microsoft Access. Avec toutes les limitations qu’il comportait, le principal problème auquel nous étions confrontés était l’impossibilité de l’interfacer avec d’autres logiciels (SAP, CRM, etc.).”
Jaime Maldonado
Responsable du laboratoire, HIPRA
“J’aime le fait qu’il soit convivial. J’aime le professionnalisme de tous mes interlocuteurs. J’apprécie également le fait qu’il s’agisse d’une amazon “STARLIMS gère de manière centralisée les échantillons et rationalise les processus de laboratoire associés pour nous permettre de fournir des données de qualité analytique en temps réel et a considérablement amélioré notre capacité à servir de point de contrôle de service pour tous les carburants aérospatiaux et les produits connexes pour les systèmes d’armement de l’AF dans le monde entier.”
Dave Fisher
Chef de la division laboratoire, Agence pétrolière de l'armée de l'air
“La solution LIMS est connectée à nos systèmes de production et de réception des matériaux au stockage. Cela signifie une avancée fondamentale dans les processus de contrôle de la qualité de l’entreprise, ainsi qu’un plus grand échange d’informations entre les systèmes.”
David Martínez Baños
Département informatique
“La plupart des systèmes sont au même niveau en ce qui concerne les fonctionnalités. STARLIMS s’est distingué en proposant un système très ouvert avec une grande flexibilité et le meilleur mélange entre les fonctionnalités disponibles et la convivialité. De plus, nous avions confiance en leur équipe de projet.”

